Bioequivalence AI Platform — From Clinical PDFs to Regulatory-Grade Assessment
a UK-based
Our client is a pharmaceutical technology company developing tools to accelerate and de-risk early-stage drug development. They had a scientifically validated hypothesis and a clear market opportunity - what they needed was a production-grade system that could make it credible in front of both technical evaluators and financial stakeholders.
1. Requirement
1.1. Purpose
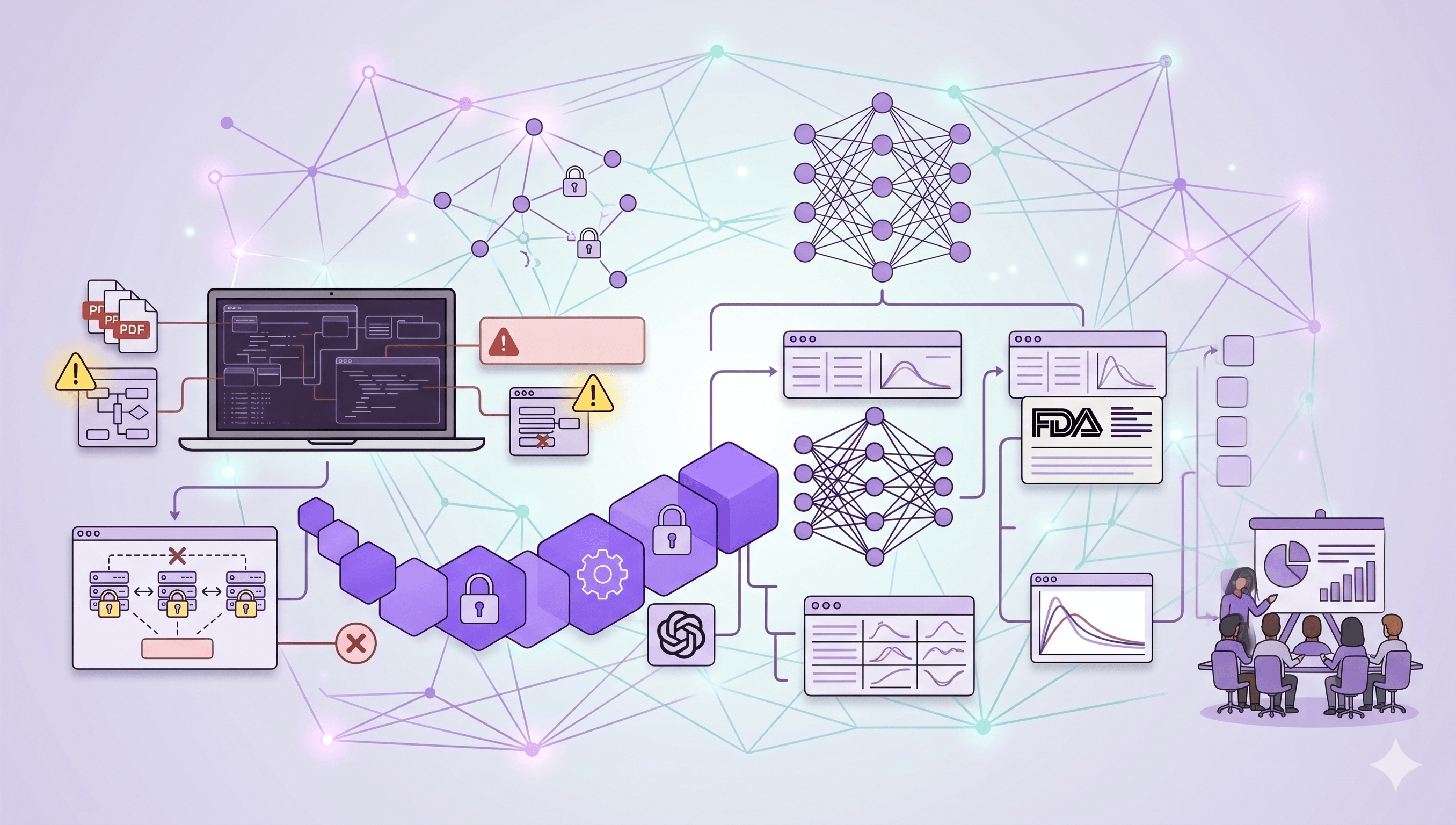
To design and engineer an enterprise-grade Bioequivalence AI Platform within a sprint. The system had to ingest unstructured clinical PDF reports, extract pharmacokinetic metrics, and generate FDA-aligned bioequivalence assessments - delivered through a production-ready web application the client team could operate independently at any scale. The platform was built to production standards from day one, with investor validation as a built-in capability, not an afterthought.
1.2. Detail Requirement
The primary goal was to deliver a system that performs correctly on real-world clinical data - not curated examples - and produces outputs that are defensible to both pharma-literate technical reviewers and financial stakeholders.
Functional Requirements
- Automated ingestion of unstructured clinical reports with no manual data entry.
- Reliable extraction of pharmacokinetic metrics and critical covariates across variable document formats.
- In-silico bioequivalence prediction with biological fidelity across standard dosage scenarios.
- Regulatory-aligned output producing definitive assessment decisions based on established FDA criteria.
- Production web application presenting results through structured regulatory output and dynamic visualizations.
Technical & Integration Requirements
- Hybrid AI architecture combining document intelligence and neural network prediction layers.
- Automated unit normalization ensuring training consistency across heterogeneous source documents.
- External data enrichment supplementing clinical feature vectors with physicochemical properties.
- Enterprise-grade cloud infrastructure with encryption at rest, audit logging, and network isolation.
Compliance Requirements
- Platform architecture mirrors HIPAA-compliant production environment standards.
- Prediction logic validated for oral solid dosage forms in Phase 1.
2. Customer Problems
- Scientific credibility at limited data volume: The client needed to demonstrate meaningful AI predictions at a stage where real-world training data is limited and heterogeneous. A system that required clean, standardized input would immediately fail on real clinical PDFs — defeating the purpose entirely.
- Zero tolerance for implausible outputs: A platform that produces even one biologically implausible result destroys credibility with pharma-literate evaluators. The system had to behave like a production bioequivalence tool across all inputs, not just controlled examples.
- Full operational independence at handoff: The client needed to run the platform independently, on-demand, without vendor presence. Any architecture requiring ongoing technical support was a failure condition, not a delivery.
1. Programming Language
- Python
2. Framework
- Streamlit
- PyTorch
- FastAPI
3. Third Party
- Third Party: AWS Textract, Large Language Model
- Infrastructure: AWS Private VPC, AWS KMS, CloudTrail, EC2 · RDS
4. Database
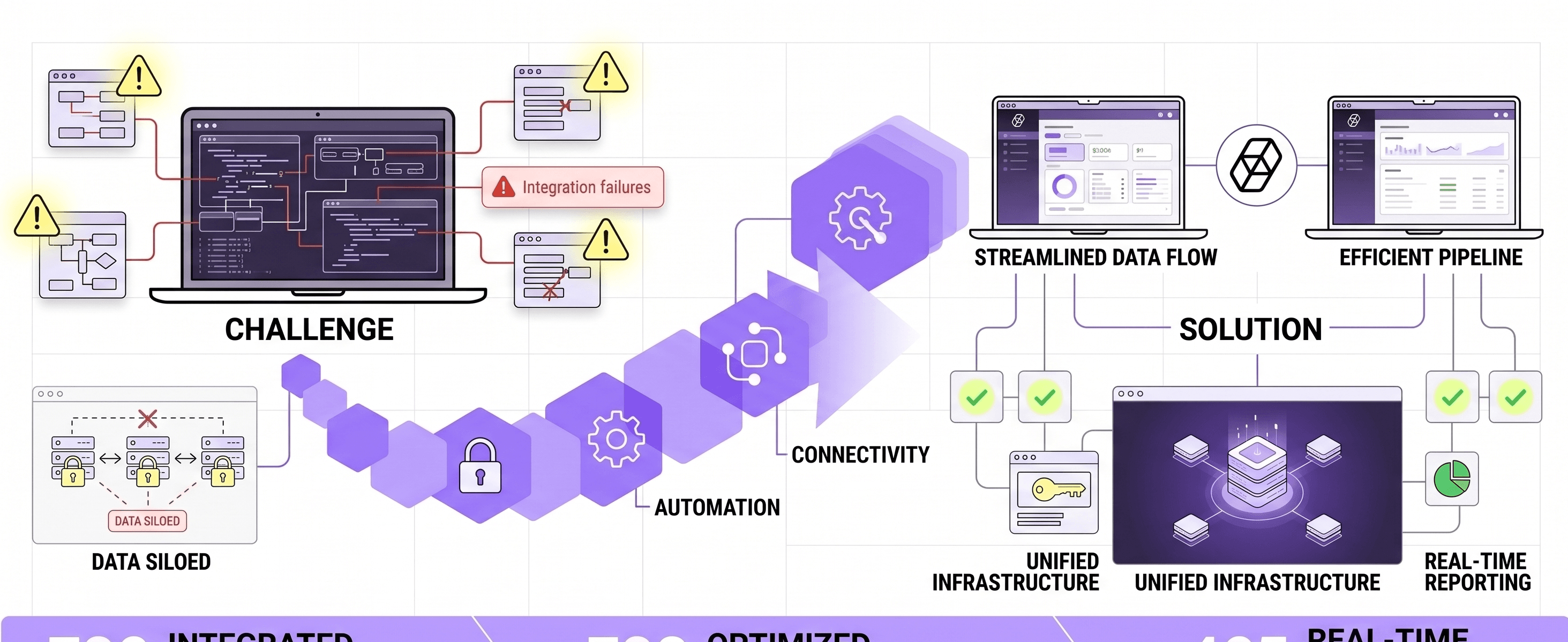
1. Challenge
Building a production bioequivalence AI platform in a sprint required solving two constraints simultaneously - neither could be traded off against the other:
- Biological fidelity under data scarcity: The prediction model had to produce results with logical alignment to reference pharmacokinetic standards - using synthetic data augmentation where clinical data was scarce - without generating outputs that a pharma-literate reviewer would flag as implausible. Correctness was an engineering requirement, not a hoped-for outcome.
- Enterprise-grade reliability at handoff: The platform had to be fully operable by the client team from day one - on their own infrastructure, under their own control, with no ongoing dependency on BeevR. Any architecture requiring vendor presence to run, interpret, or maintain was unacceptable.
2. How to resolve these challenges
1. Domain constraints as architectural requirements, not guardrails. Bioequivalence prediction fails when a model treats pharmacokinetics as a generic regression problem. BeevR designed the prediction layer around known biological behaviors - not as training targets, but as hard architectural constraints. The system produces results that are defensible to pharma-literate reviewers because biological correctness was engineered in, not validated after the fact.
2. Production standards applied from sprint day one. The platform was deployed within an enterprise-grade AWS infrastructure - private network isolation, encryption at rest, full audit logging - from the first deployment, not retrofitted at the end. This meant the client received a system that was ready for technical due diligence on day 10, not a prototype requiring hardening.
3. Data constraints treated as a design input. Limited clinical data at early stage is not an exception in pharmaceutical AI - it is the norm. BeevR designed the ingestion and training pipeline to operate correctly under this constraint from day one, including automated handling of inconsistent document formats, variable reporting units, and sparse training signals, rather than deferring these as Phase 2 problems.
Delivering Results:
- Fully operational platform delivered at Week 2 as contracted — 10 business days
- Fixed price, zero scope overruns
- 2.8s median latency · 99.99% uptime during client evaluation period
- 82% automated test coverage (1,200 backend / 800 frontend / 400 tests)
- $120/month infrastructure cost post-handoff
- Full ownership transferred: source code, trained model weights, infrastructure - deployed within client's own AWS environment
- Zero ongoing dependency on BeevR
Other Works
From Prototype to Enterprise-Ready: A macOS Security Agent That Actually Ships
From proof-of-concept to enterprise distribution - a macOS compliance agent hardened, signed, and production-ready.

From Reactive to Predictive - Revolutionizing US B2B Services
Revolutionizing US B2B Services from Reactive to Predictive using AI/ML and Custom ERP; achieving 15% cost savings and 30% process optimization.

Optimizing Manufacturing Assembly with Pick-to-Light
A software solution that transforms manual parts-picking into a fast, accurate, and efficient process for modern manufacturing assembly lines.
